Chroniques génomiques - Immunothérapie « CAR-T » : une autorisation qui fait date
Résumé
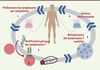
The first therapy based on chimeric antigen receptors has been approved by the FDA. It gives excellent results in recurrent paediatric ALL; its cost is very high but fairly consistent with the clinical benefit (long term remission approaching a cure). In spite of their complexity and cost, CAR-T therapies will probably end up playing a very significant role in cancer therapy.
Pour citer ce document
Jordan, Bertrand ; Chroniques génomiques - Immunothérapie « CAR-T » : une autorisation qui fait date, Med Sci (Paris), , Vol. 33, N° 11 ; p. 1003-1006 ; DOI : 10.1051/medsci/20173311020